It’s important for flu vaccine providers to continue to educate the public on the importance of receiving vaccines, as well as ensuring your vaccine supply is safely stored. Proper vaccine storage can prevent waste and financial loss due to unwanted temperature excursions. Following CDC recommendations for the safe storage of vaccines, including the flu vaccine, helps ensure that every flu vaccine dose counts. This is key to keeping flu cases as low as possible.
Read on to find the answers to the most commonly asked questions about flu vaccine refrigeration to help you ensure an effective immunization program at your practice.
Frequently Asked Questions about Flu Vaccine Refrigeration
What type of refrigeration unit do you need to store flu vaccines?
The ideal units for flu vaccine refrigeration are pharmacy-grade or stand-alone refrigerators. Household combination units may be used in some cases, but dormitory-style units should never be used. The best case scenario is a refrigerator that is purpose-built for storing vaccines.
Can the flu vaccine be stored at room temperature?
No. The flu vaccine must be stored in a refrigerator unit at a temperature between 35° and 46°F (2 and 8°C). The vaccine should never be frozen or kept outside of a refrigerator.
What if the flu vaccine is not refrigerated?
Flu vaccines become less effective and potentially unsafe if they are not refrigerated, have been frozen, or have been affected by a temperature excursion event. Do not administer these vaccines to patients. Instead, document the issue and contact the vaccine manufacturer or your state immunization program for guidance.
Can a combination unit to store all our vaccines together, with flu vaccines in the fridge and COVID-19 vaccines in the freezer?
Combination units are not recommended for storing vaccines as they do not ensure even temperatures in both compartments. Do not store vaccines in both the refrigerator and freezer compartments.
The CDC recommends that you only store vaccines in the refrigerator compartment of a combination unit. Do not turn off the freezer compartment as this may also affect the refrigerator temperature. Many combination units use the cold air from the freezer to cool the refrigerator. Frozen vaccines should be kept in a separate vaccine freezer unit. Before choosing another unit, be clear on the exact temperature requirements of other vaccines, especially in the case of COVID-19 vaccines.
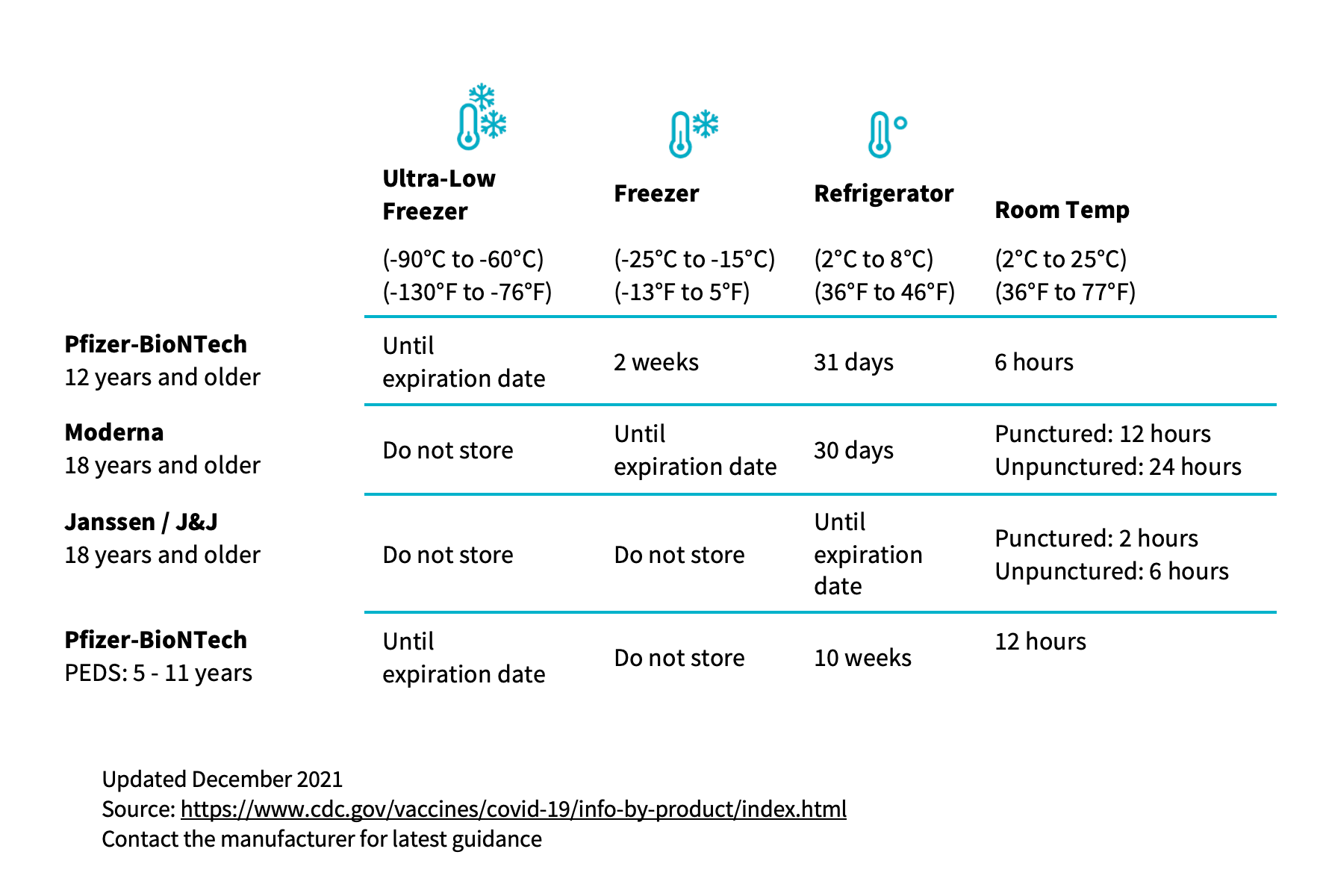
COVID-19 vaccines require different temperature ranges depending on the product. These temperatures vary from refrigerated (between 2 and 8°C) through frozen (between -15° and -25°C) to ultra-cold (between -60° and -80°C). As with most vaccines, COVID-19 vaccine storage temperature is essential for vaccine efficacy. Do not store COVID-19 vaccines in a combination unit with other vaccines and make sure any other storage unit meets the correct temperature requirements for the product.
Can biological products be stored in the same refrigerator with flu vaccines?
No. The frequent opening and closing of the refrigerator door as you access other items can cause serious temperature excursions. This risks flu vaccine efficacy. Your refrigerator unit should not be overused for this reason. Store biological products in a separate refrigerator, to ensure the safety of your vaccine supply.
Can the top shelf of the refrigerator be used to store flu vaccines?
No. Do not store flu vaccines on the top shelf of the refrigerator. Temperatures can vary widely, especially if there is a freezer compartment above the refrigerator with a cold air vent. Always store flu vaccines in the middle of the unit, where the air can circulate and temperatures are the most even.
Unlike household units, pharmacy-grade refrigerators have a fan that circulates the air to ensure more consistent temperatures. Even with these specialized units, you should still avoid using the top shelf to store flu vaccines, as temperatures rise most quickly in this part of the refrigerator during power outages.
How often should we log refrigerator temperatures when storing the flu vaccine?
When storing any vaccine, you should log the minimum and maximum refrigerator temperatures at the beginning and end of each day. If the unit does not display the minimum and maximum temperatures, log the current temperature at the beginning and end of each day.
You can also measure the temperature of the room where the refrigerator is kept. In the event of a temperature excursion or refrigerator failure, you will have a better idea of the temperature your vaccines have been exposed to, which can help with the troubleshooting process. Do not use the calibrated temperature monitor from the refrigerator to measure the room temperature, but rather a standard household thermometer.
How long should we keep flu season temperature logs for?
The CDC recommends keeping all your temperature logs for three years. If you are participating in your state’s Vaccines for Children program (VFC), you may be required to retain and provide your temperature logs. By keeping this information, you will also be able to trace any issues with your general vaccine and flu vaccine refrigeration methods. Check with your state or local program to understand your temperature logging requirements.
What steps do we need to take if the refrigerator has an excursion?
As soon as you discover an excursion event, label all of the inventory as “Do Not Use.” You should report the incident to clinic staff and fill out any vaccine storage troubleshooting records used at your practice. Contact the vaccine manufacturer or your local or state health department to determine whether the affected vaccines can still be safely administered to patients or should be disposed of.
Your flu vaccine refrigeration unit should be restored to its proper conditions and the reasons for the temperature excursion should be identified as quickly as possible. If your refrigerator cannot maintain the recommended temperature range for vaccine storage, you should use another unit with more reliable temperature control.
How can I ensure proper Flu and Covid-19 Vaccine Storage?
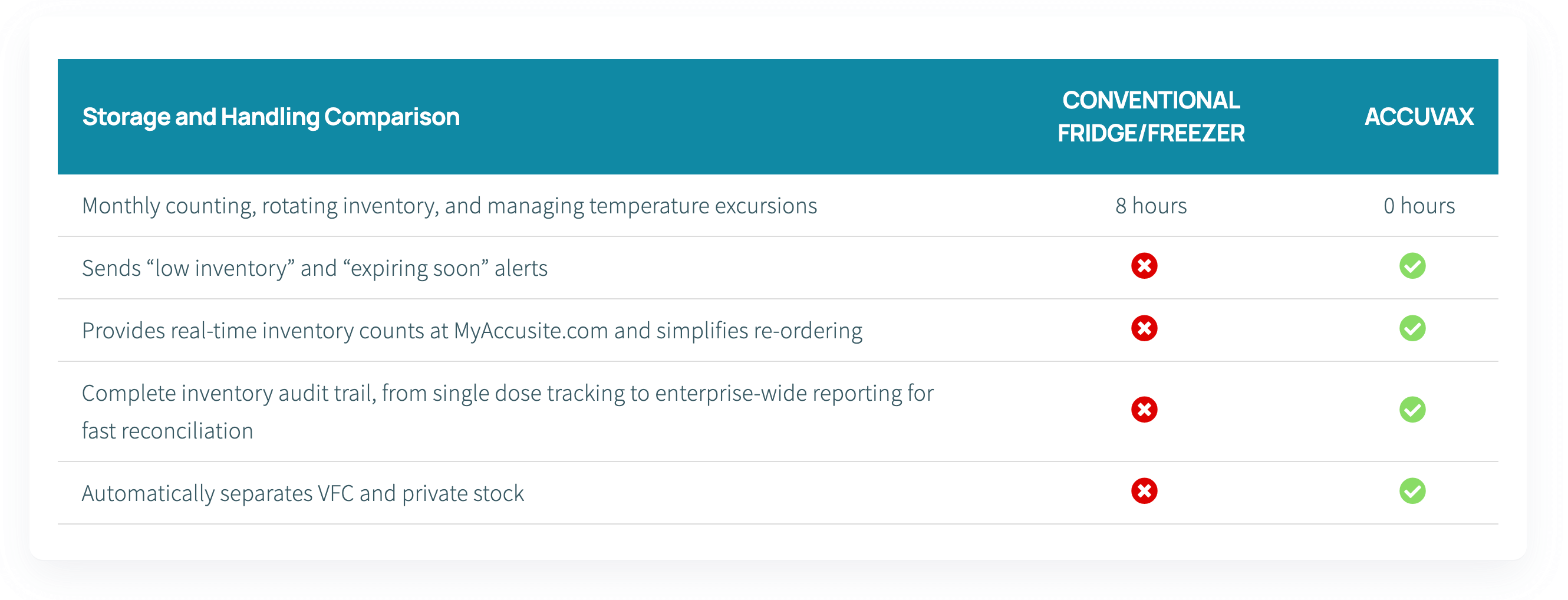
Even if your practice carefully monitors flu vaccine refrigeration and COVID-19 vaccines, temperature excursions can still occur and damage your vaccine inventory. Only a compliant storage system can remove the costly risks of human error and ineffective equipment. With guaranteed temperature control and constant remote monitoring, the AccuVax Vaccine Management System will keep your flu vaccines and COVID-19 vaccines at the ideal temperature, without any manual organization or tiresome data-logging.
Unlike dangerous dormitory-style units and unreliable combination units, AccuVax can safely store both refrigerated and frozen vaccines in one unit. The purpose-built, pharmacy-grade system automatically separates vaccines, so you don’t need to worry about storing them incorrectly and letting them go to waste. What’s more, the door-less technology and built-in battery backup eliminate the main causes of temperature excursions and guarantees the protection of all vaccines stored in the AccuVax.

AccuVax workflows are designed to simplify all areas of vaccine storage & management, allowing staff and the entire practice to focus on higher-value care. Prevent staff, clinicians, and doctors from time-consuming vaccine management tasks with the AccuVax solution:
Find out how the AccuVax can support your practice through the busy flu season to provide an effective immunization program.