Your practice cannot afford to let vaccines go to waste. Coronavirus (COVID-19) vaccines are in short supply. If your inventory falls out of the required vaccine storage temperatures, vaccines can become damaged, ineffective, and unsafe to use. This can slow down your immunization program and put your patients at risk (see below for a simple vaccine storage temperature chart).
Storing and managing different vaccines at different temperatures only adds another challenge to think about. Current COVID-19 vaccine temperature ranges are much colder than what you may be used to working with. Manual processes prevent your practice from focusing on patient care and can result in human error. Even with a staff their to help you keep an extra eye on this inventory many problems can arise.
Related: What You Need to Know: Flu Vaccine Refrigeration
Read on to learn about storage temperatures of the authorized COVID-19 vaccines. We’ll also talk about how you can protect your valuable inventory until your patients are ready and able to receive a vaccination.
Why Temperature Is Key…
The COVID-19 vaccines need to be stored at specific temperatures to maintain their efficacy. Unpunctured Johnson & Johnson vials can be safely refrigerated for 3 months. Longer-term storage temperatures for Pfizer-BioNTech and Moderna vaccines need to be much colder. These vaccines contain messenger RNA (mRNA), a genetic material found in all cells used to create proteins. MRNA requires careful temperature monitoring.
Your body uses the mRNA to create its own version of the spike protein. The spike protein is a component of the outer shell of the COVID-19 virus which triggers an immune response in your body. Your immune system learns to fight any future exposure to the virus from spike protein triggers.
MRNA vaccines were the first to be authorized because they can be synthesized in a laboratory without a large production of the virus. However, mRNA is a fragile material that breaks down quickly and easily. Cold vaccine storage temperatures are required to keep the mRNA stable until it’s ready to be used.
Future COVID-19 vaccines may not require such extreme temperature ranges, especially if they do not contain mRNA. Nevertheless, it’s critical to adhere to all the different vaccine storage temperatures to keep vaccines safe and effective.
Why Does the Pfizer-BioNTech Vaccine Require Colder Storage Temperatures Than the Moderna Vaccine?
The Pfizer-BioNTech vaccine needs an ultra-cold chain resulting in logistical challenges of shipping and storing. But there are good reasons for these ultra-cold vaccine storage temperatures.
Pfizer-BioNTech had to ensure vaccine stability. The manufacturer went with the safest option: recommending an ultra-cold vaccine temperature range. With more trials, the required vaccine storage temperatures may change. In fact, the FDA has recently allowed more flexible storage for the Pfizer-BioNTech vaccine.
Although there may be more changes in the future, it’s important to maintain the vaccine temperature ranges that are currently recommended. Even though the proteins of both the Pfizer-BioNTech and Moderna vaccines were derived from the same material, they are not the same and may still require very different conditions.
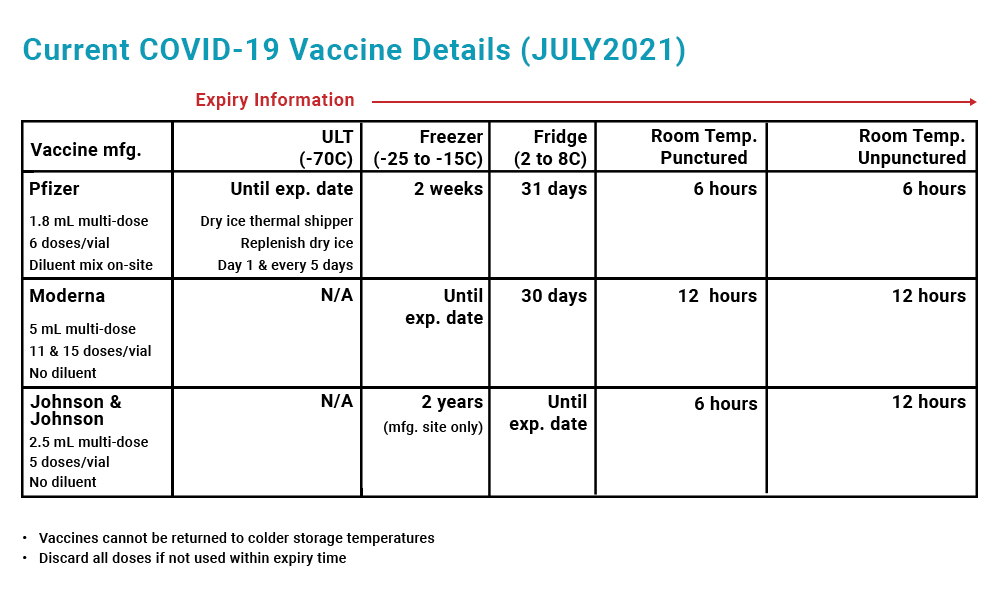
COVID-19 immunization guidelines for Pfizer, Moderna, and Johnson & Johnson.
COVID-19 Vaccine Temperature Ranges
As each manufacturer has created a COVID-19 vaccine that has different temperature requirements, it’s essential to maintain the specific storage temperatures for the vaccine your practice is using. The different manufacturers are not interchangeable and temperature is key to vaccine efficacy.
You should not store COVID-19 vaccines in a combination unit with other vaccines that require different storage temperatures. If any vials fall out of these vaccine temperature ranges, do not use them. Dispose all vaccines if they are not used within their expiry times or have been affected by a temperature excursion event at any point during storage.
Pfizer-BioNTech Vaccine Storage Temperature
The Pfizer-BioNTech vaccine can be stored between -80 and -60°C until the expiration date. If you keep the vaccines in the dry ice thermal shipper, replenish the dry ice on day 1 and day 5 of the storage period. You can also store this vaccine in a freezer unit between -25 and -15°C for up to 2 weeks.
This vaccine needs to be thawed and reconstituted before use. You can thaw the vials in a refrigerator unit between 2 and 8°C for 3 hours, or at room temperature (up to 25°C) for 30 minutes. Once the vaccines have been thawed and reconstituted, they cannot be refrozen. They can, however, stay safely in the refrigerator for up to 31 days.
Store punctured vials in a refrigerator unit between 2 to 8°C for up to 5 days or at room temperature (up to 25°C) for no more than 6 hours to prevent bacterial contamination.
Moderna Vaccine Storage Temperature
The Moderna vaccine can be stored in a freezer unit between -25 and -15°C until the expiration date. You can also store the vaccine in a refrigerator unit between 2 to 8°C for up to 30 days.
If stored in a freezer unit, this vaccine needs to be thawed before use. You can thaw the vials in a refrigerator unit between 2 and 8°C for 2 hours and 30 minutes, or at room temperature (up to 25°C) for 1 hour. Once the vaccines have been thawed, they cannot be returned to the freezer.
Store punctured vials in a refrigerator unit between 2 to 8°C for up to 30 days or at room temperature (up to 25°C) for no more than 12 hours to prevent bacterial contamination.
Johnson & Johnson Vaccine Storage Temperature
The Johnson & Johnson vaccine can be stored in a refrigerator unit between 2 to 8°C until the expiration date. Once the vials have been punctured, they can be stored in the refrigerator for up to another 6 hours. After this time, if the vaccines have not been used, they must be discarded.
This vaccine can also be stored at room temperature for up to 12 hours before puncture and for up to 6 hours after puncture.

How Your Practice Can Protect Vaccines
A vaccine storage and monitoring system you can rely on is crucial during this busy immunization boom. Ensure that you keep your vaccines safe and effective until you’re able to administer them to your patients. The following technology can help you protect your vaccines by ensuring the correct vaccine storage temperatures.
Vaccine Temperature Monitoring
Manual vaccine temperature monitoring requires time and vigilance. A member of your team needs to log temperatures in the morning and evening each day. COVID-19 vaccine temperature ranges are more larger and they are critical to efficacy. Monitoring has never been more important.
Temperature excursion events can still take place between these daily check-ins and lead to even more manual action. All out-of-range temperatures need to be logged and looked into. Your team member may also need to contact the vaccine manufacturer or local or state health department to determine whether the vaccines can still be used.
The AccuShelf Inventory Management System provides integration with digital data loggers which can be attached to any cold storage device, including dry ice containers and ultra-low freezers. Ultra-low and standard vaccine temperature monitoring allows you to maintain the correct COVID-19 vaccine storage temperatures and track movement between storage units. With real-time temperature alerts, you can react quickly to any temperature excursions that might put your vaccines at risk.
Vaccine Refrigerator with Battery Backup
Temperature excursion events can stem from poor quality storage equipment, a team member accidentally leaving the storage unit door open, or from power outages. Whatever the cause, your inventory falls out of the vaccine temperature ranges and may have to be discarded.
In addition to its doorless design that prevents human error, the Accuvax Vaccine Refrigerator with Battery Backup provides up to 15 hours of vaccine storage protection. The built-in battery pack ensures your vaccines stay at the correct vaccine storage temperatures to give you extra peace of mind during this stressful time.
Purpose-Built Vaccine Storage
Household combination storage units are often unable to reach and maintain COVID-19 vaccine storage temperatures. These devices have both a freezer and refrigerator compartment, but they cannot store a variety of vaccines at different temperature ranges. What’s more, all stock loading and rotation is done by hand.
Store vaccines safely and effectively so they are easily available and used before their expiration. A purpose-built vaccine storage unit that can support your entire inventory. No matter how many different vaccine storage temperatures you need to accommodate. A custom-built vaccine storage system allows you to scale up when medical needs change and expand.
As the emerging standard in the industry, the AccuVax Vaccine Management System can store refrigerated and frozen vaccines safely. AccuVax keeps them within the same unit and guarantees temperature control. The technology automatically rotates your inventory by expiry date to prevent waste. Notifications and real-time dose level data inform you so you know exactly how to manage your vaccines. Provide your patients with effective vaccinations when they need it.
Schedule a demo today to learn how TruMed Systems can help you protect your COVID-19 vaccines for a more effective immunization program at your practice.